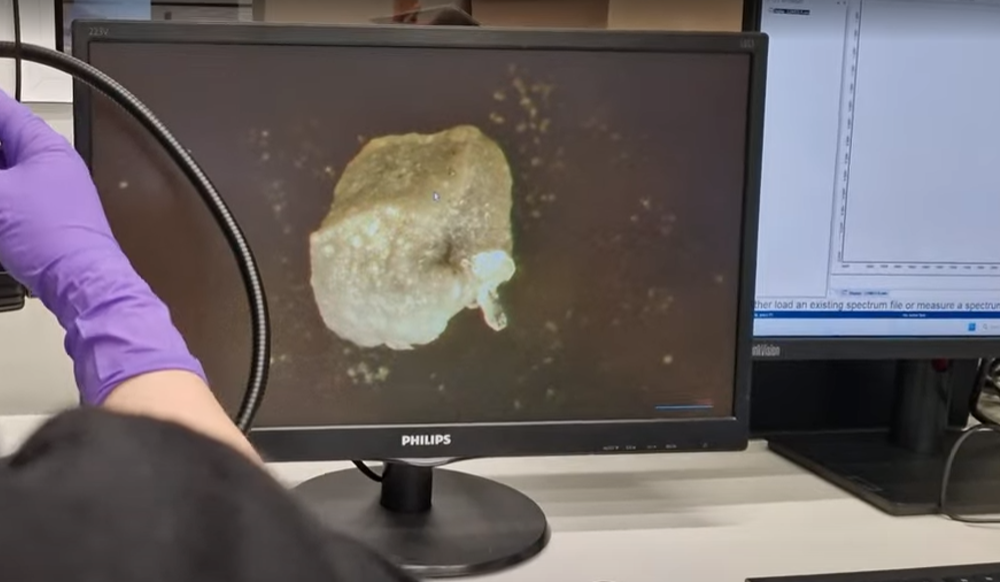
Particle Identification with SEM & FTIR
eyetec helps pharmaceutical manufacturers identify unknown particles.
Using Scanning Electron Microscopy (SEM) and Fourier Transform Infrared (FTIR) analysis, we determine what the particle is made of. This insight helps teams trace the source of contamination. As a result, corrective actions can be implemented quickly and the risk of recurring contamination is reduced.
Detected a particle.
But unsure what it is or where it comes from?
During our Particle Insight Program, you can submit a particle for review. Submissions are open until 31 May.
Submit your particle
Combining SEM and FTIR
Our particle identification service combines the strengths of Scanning Electron Microscopy (SEM) and Fourier Transform Infrared (FTIR) analysis.
These two analytical techniques provide different but complementary information about a particle.
SEM focuses on the structure and elemental composition of the particle.
FTIR focuses on the molecular structure of organic materials.
By combining both techniques, we can analyze a wide range of materials and obtain reliable particle identification results.

Scanning Electron Microscopy (SEM)
SEM uses a focused beam of electrons to scan the surface of a particle. This produces high-resolution images of the particle.
SEM allows us to study:
• particle morphology (shape and surface structure)
• particle size and surface features
• elemental composition through EDX analysis
Elemental analysis identifies which chemical elements are present in the particle.
This makes SEM particularly suitable for inorganic materials, such as:
• metals
• metal alloys
• glass fragments
• mineral particles

Fourier Transform Infrared (FTIR) analysis
Fourier Transform Infrared Spectroscopy
FTIR identifies materials based on their molecular structure.
The technique exposes the sample to infrared light.
Molecules absorb this light at specific wavelengths.
This produces a unique spectral fingerprint for each material.
FTIR allows us to identify organic materials, such as:
• polymers
• plastics
• rubber components
• fibers
• packaging materials
Comparing with production materials
When investigating contamination, identifying the particle alone is often not enough.
To support the investigation, we can also analyze reference samples from the production process.
These may include materials such as:
• equipment components
• packaging materials
• process materials
• environmental sources
By comparing these reference samples with the detected particle, we can determine where contamination most likely occurred.
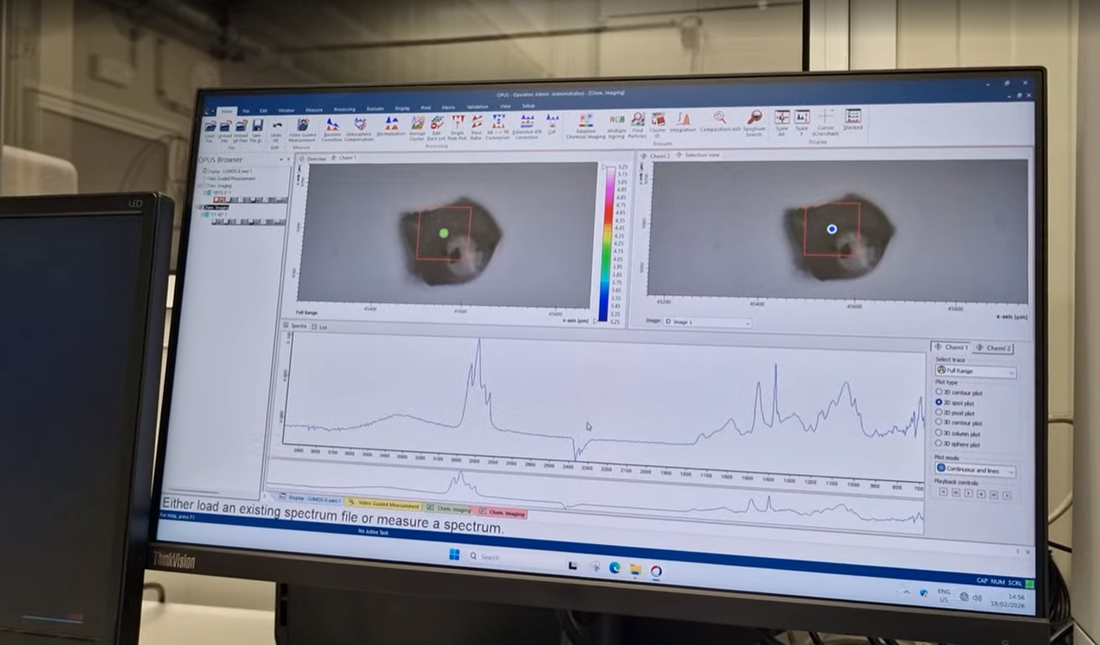
From particle identification to process improvement
Understanding the origin of contamination allows quality teams to:
• implement targeted corrective actions
• prevent recurrence
• improve process reliability
Particle identification therefore does more than resolve individual incidents.
It also supports continuous improvement of the production process.
Particle identification with SEM and FTIR
